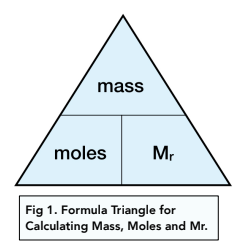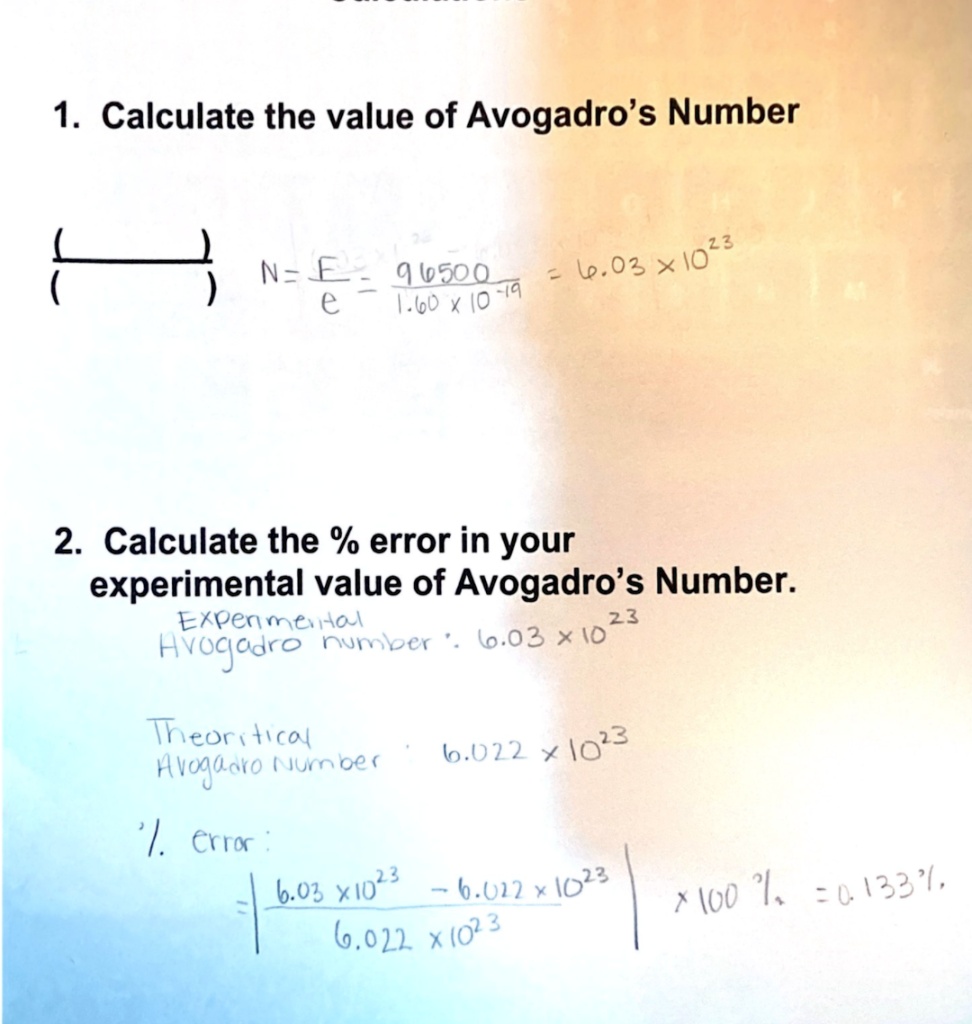
SOLVED: 1 Calculate the value of Avogadro's Number 13 l.03 x10 1 N= € 9.k5oo A.6o / I0 Fiq 2. Calculate the % error in your experimental value of Avogadro's Number: Ekpen

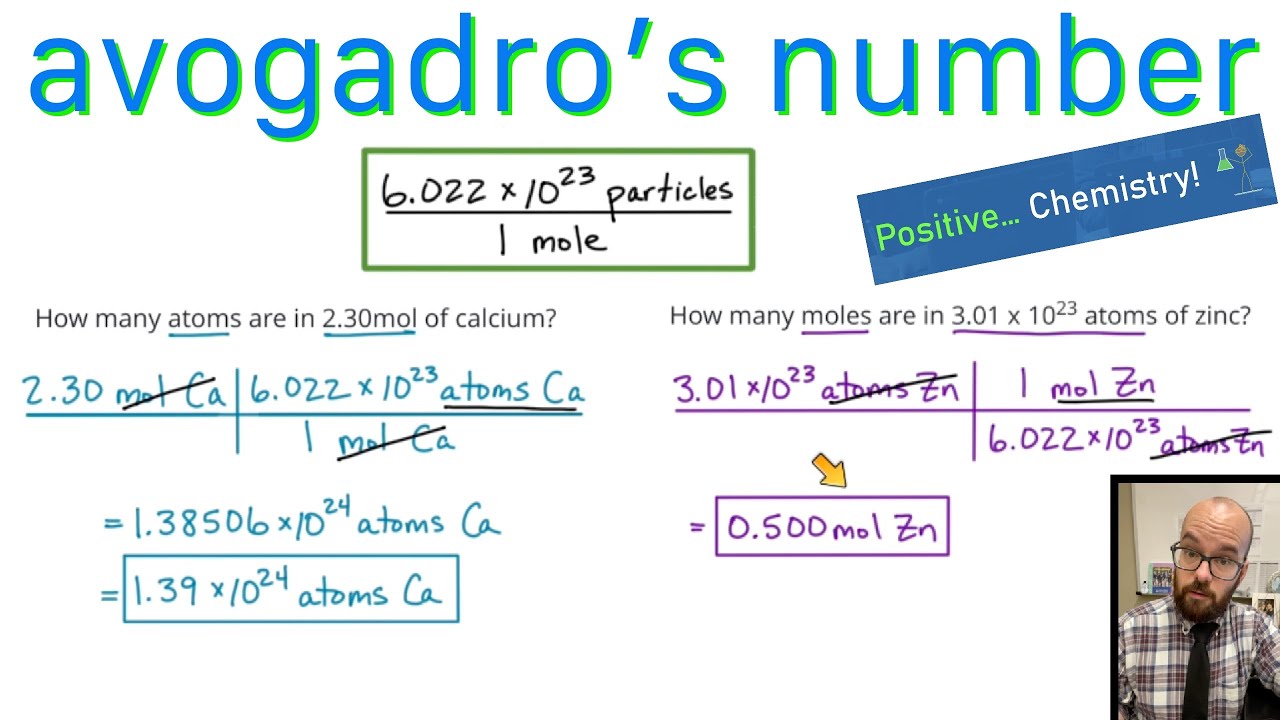
1. Using Avogadro's number, calculate the number of atoms in 0.005 kilograms of carbon. 2. If there are 'x' atoms in 5 grams of carbon, how many atoms are there in 5

Calculate the value of Avogadro's number from the following data: Density of NaCl = 2.165 g cm^(-3) Distance between Na^(o+) and Cl^(Θ) in NaCl = 281 pm

Physical Chemistry #1: Relative Mass, the Mole and Avogadro's Constant (Slides & Student Led Tasks) | Teaching Resources