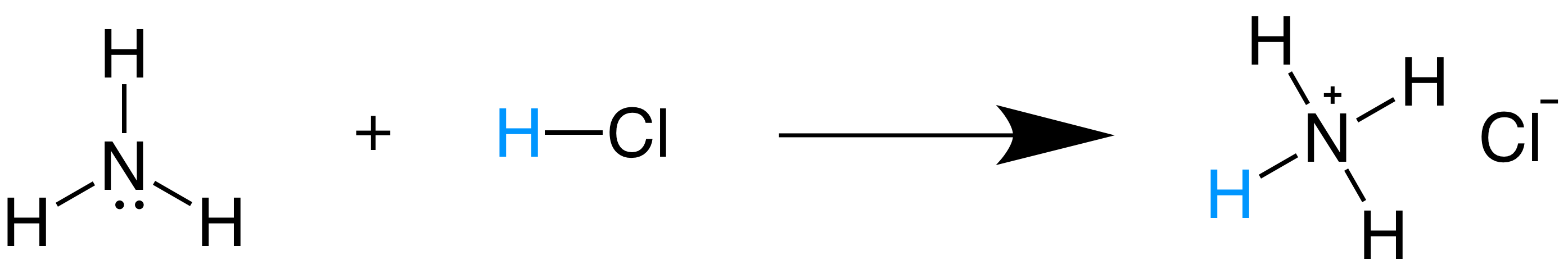SOLVED: Identify the conjugate acid-base pairs in the reaction shown below HBr + NH3 Br + NH4 acid; chemPad Help Gianke conjugate base: chemPad Help Girekt base: chemPad Help Creke conjugate acid:
Which pair is a Brønsted–Lowry conjugate acid–base pair? NH_3 ; NH_4^+ or H_3O^+ ; OH^- or HCl; HBr or ClO_4^(-); ClO_3^- | Socratic
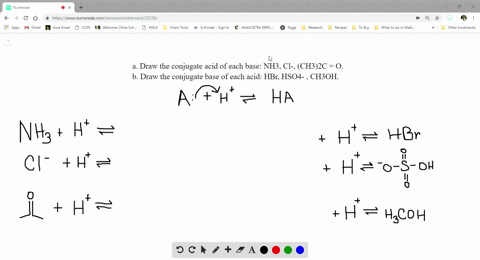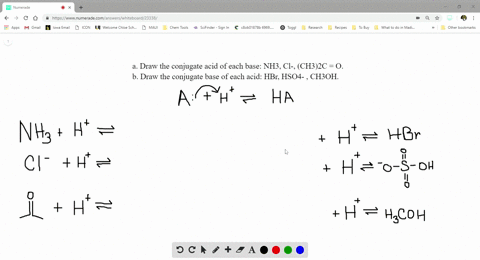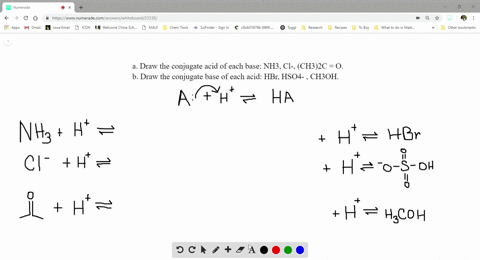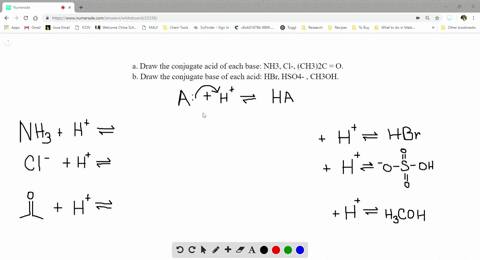
SOLVED:a. Draw the conjugate acid of each base: NH3, Cl^-, (CH3)2C = O. b. Draw the conjugate base of each acid: HBr, HSO4^- , CH3OH.

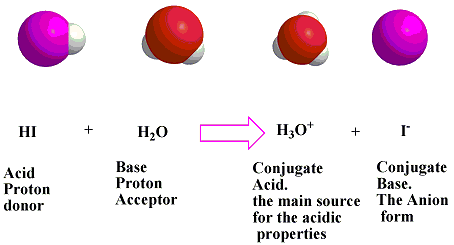
PHILIPPINE CHEMISTRY PROFESSIONALS BOARD EXAM REVIEWER - Which one of the following mechanistically depicts the acid-base reaction that occurs when hydrobromic acid(HBr) is added to methanol (CH4O)? Please refer to attached image.

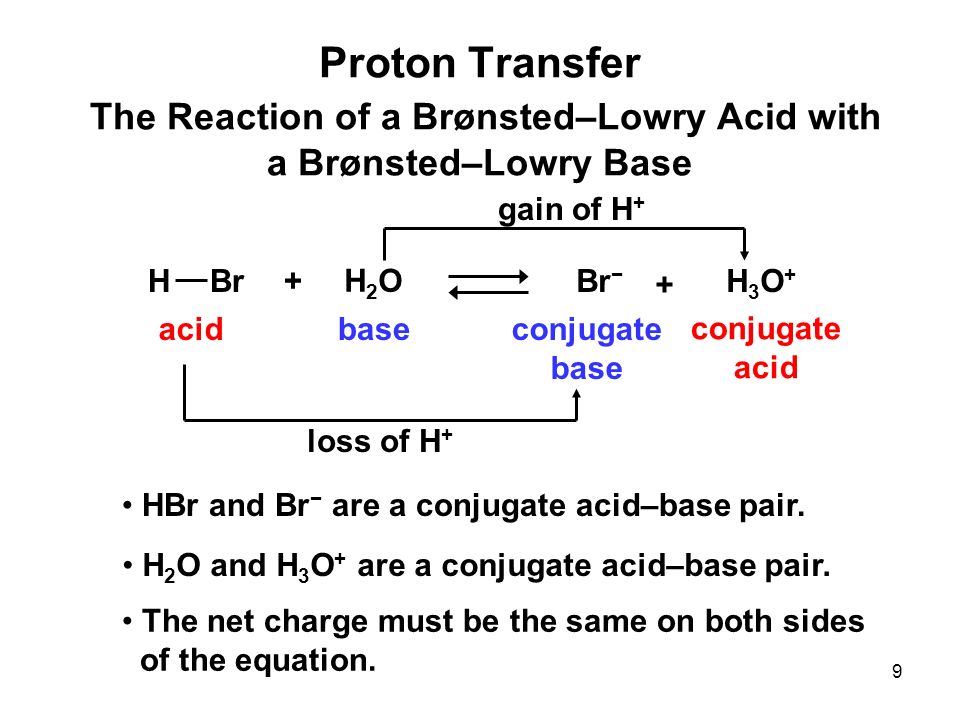
PPT - HBr is a strong acid , so the reaction goes to completion. PowerPoint Presentation - ID:4536389

PPT - HBr is a strong acid , so the reaction goes to completion. PowerPoint Presentation - ID:4536389
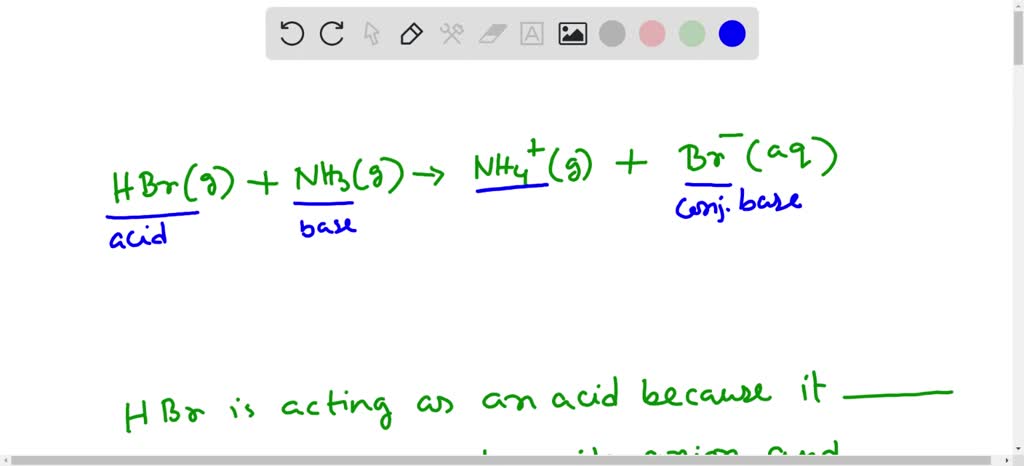
SOLVED: In the following acid-base reaction: HBr(g) + NH3(g) → NH4+(aq) + Br– (aq) HBr is acting as the acid, because it a proton to form bromide anion, and NH3 is acting