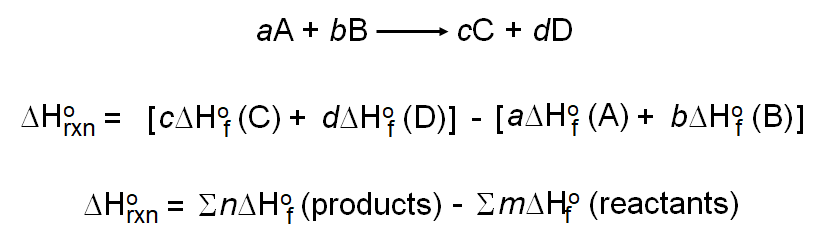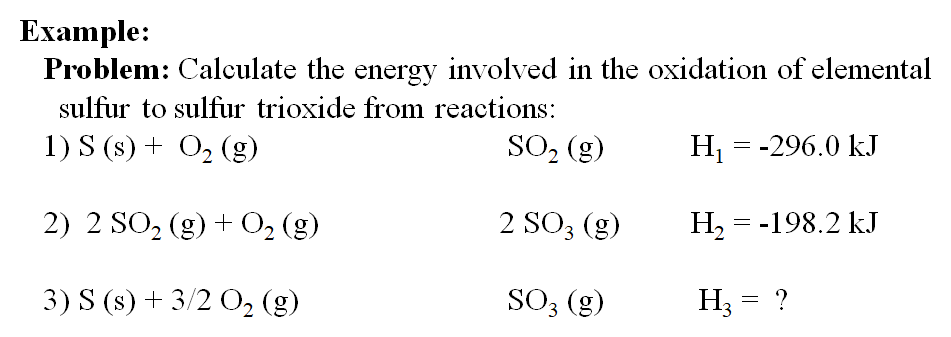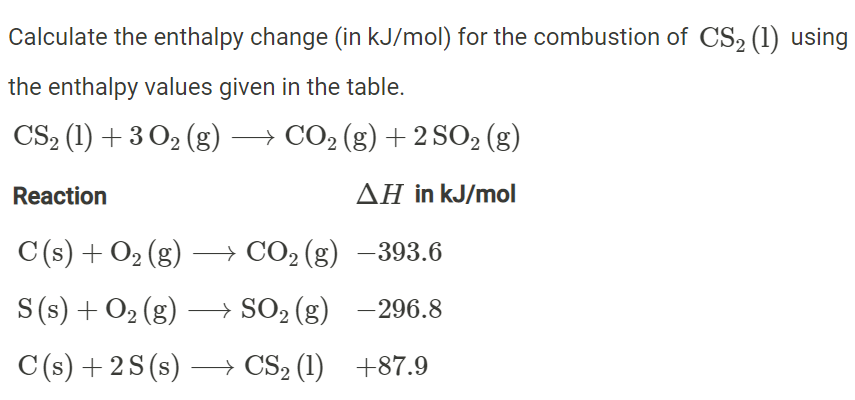Calculate the enthalpy for the reaction, H2O (g) ⟶ 2H (g) + O (g) and hence, calculate the bond enthalpy of O - H bond in H2O from the following data: ΔvapH (
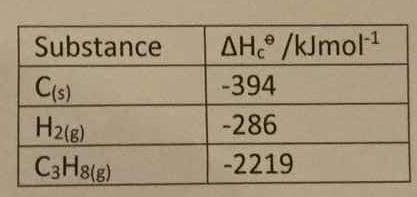
How do you determine the enthalpy change for the reaction below using the enthalpy of combustion data in the table: 3C_((s)) +4H_(2(s)) -> C_3H_(8(s))? | Socratic
![Chemistry] Does anyone know how to calculate the enthalpy change for this reaction: : r/HomeworkHelp Chemistry] Does anyone know how to calculate the enthalpy change for this reaction: : r/HomeworkHelp](https://preview.redd.it/chemistry-does-anyone-know-how-to-calculate-the-enthalpy-v0-k0t68mmrx7n81.jpg?width=640&crop=smart&auto=webp&s=76fb8b2caf8274c7660cc18d136d2a3c28bb1764)
Chemistry] Does anyone know how to calculate the enthalpy change for this reaction: : r/HomeworkHelp

Calculate the enthalpy change for the process CCl4(g)→ C(g) + 4Cl(g) and calculate bond enthalpy of C - Cl in CCl4(g) Δ vapH^ (CCl4) = 30.5 kJ mol ^-1 . Δ fH^ (

Given the chart of bond energies, calculate the enthalpy change for the reaction below. Show all work - brainly.com










![Example] How to Calculate Enthalpy Change of a Reaction. - YouTube Example] How to Calculate Enthalpy Change of a Reaction. - YouTube](https://i.ytimg.com/vi/nmNQUGt6NiM/maxresdefault.jpg)