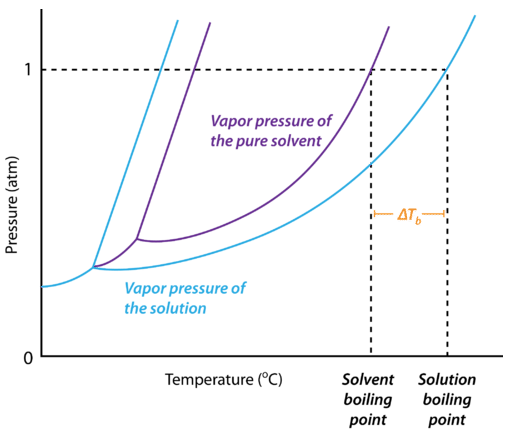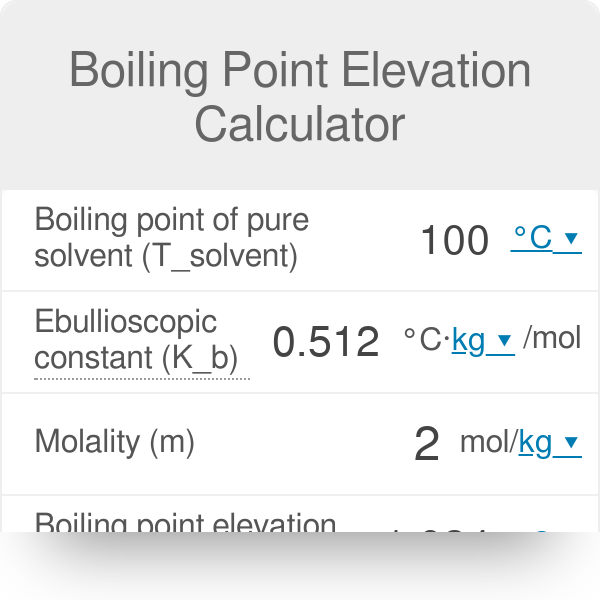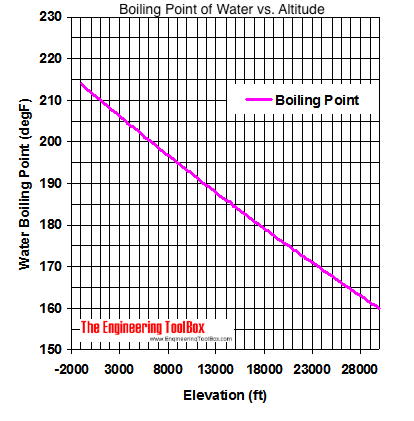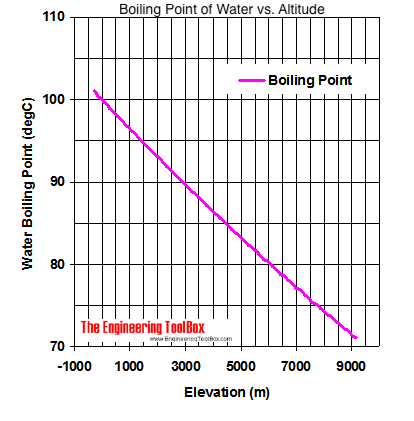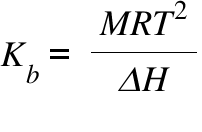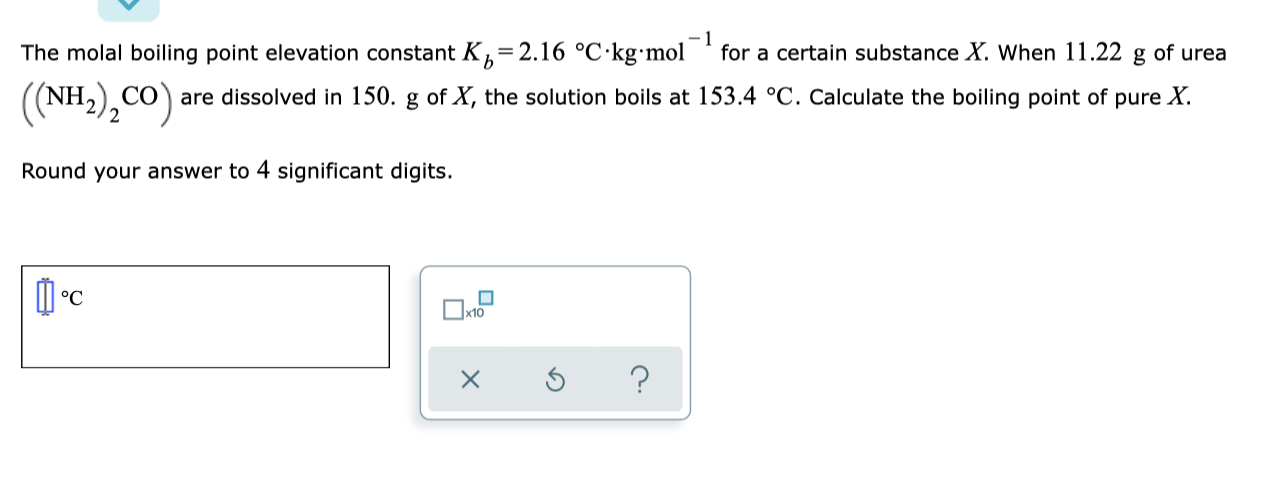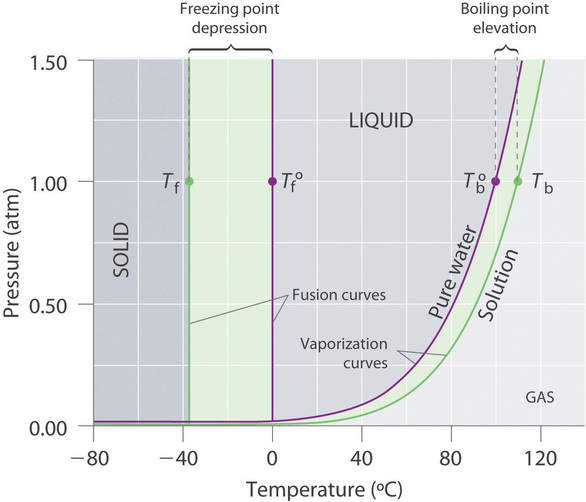
13.8: Freezing-Point Depression and Boiling-Point Elevation of Nonelectrolyte Solutions - Chemistry LibreTexts

SOLVED: The elevation in boiling point, when 0.30 g of acetic acid is dissolved in 100 g of benzene is 0.0633OC. Calculate the molecular weight of acetic acid from this data. What

Calculations Involving Colligative Properties Freezing Point Depression and Boiling Point Elevation Calculations. - ppt download
Calculate the molal elevation boiling point constant of a solution containing 1.0 gm of urea (M = 60) in 75.0 gm water and boils at - Sarthaks eConnect | Largest Online Education Community

Calculate the freezing point and the boiling point at 1 atmosphere of a solution containing 30 g cane sugar (molecular mass 342 ) and 150 g water.Given : Kb = 0.513 and Kf = 1.86

Boiling Point Overview & Calculation | How to Calculate Boiling Point - Video & Lesson Transcript | Study.com