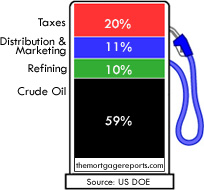
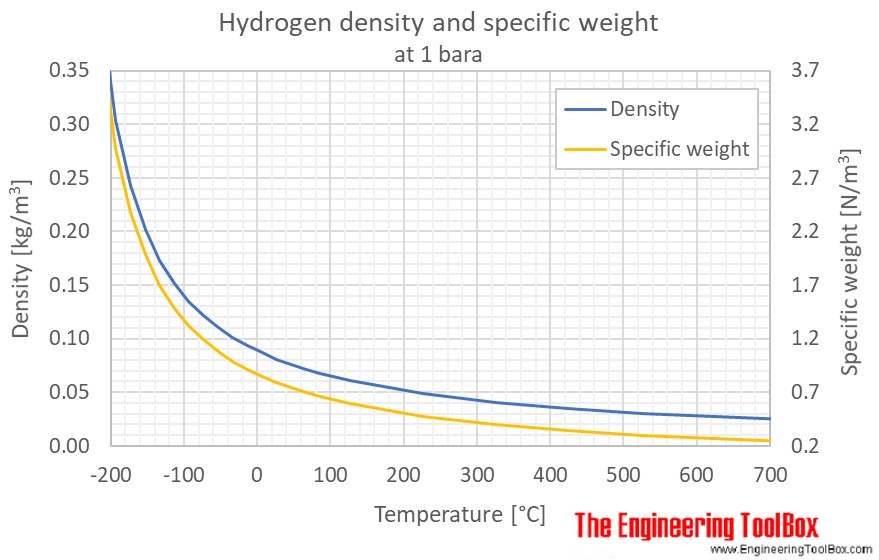
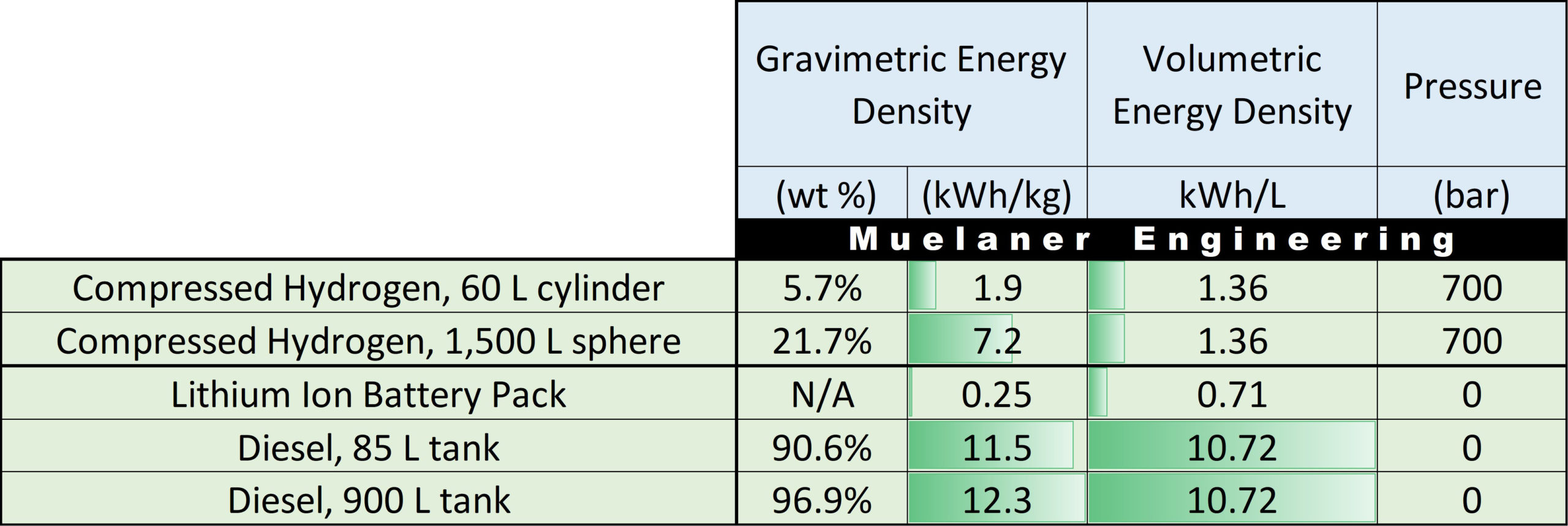
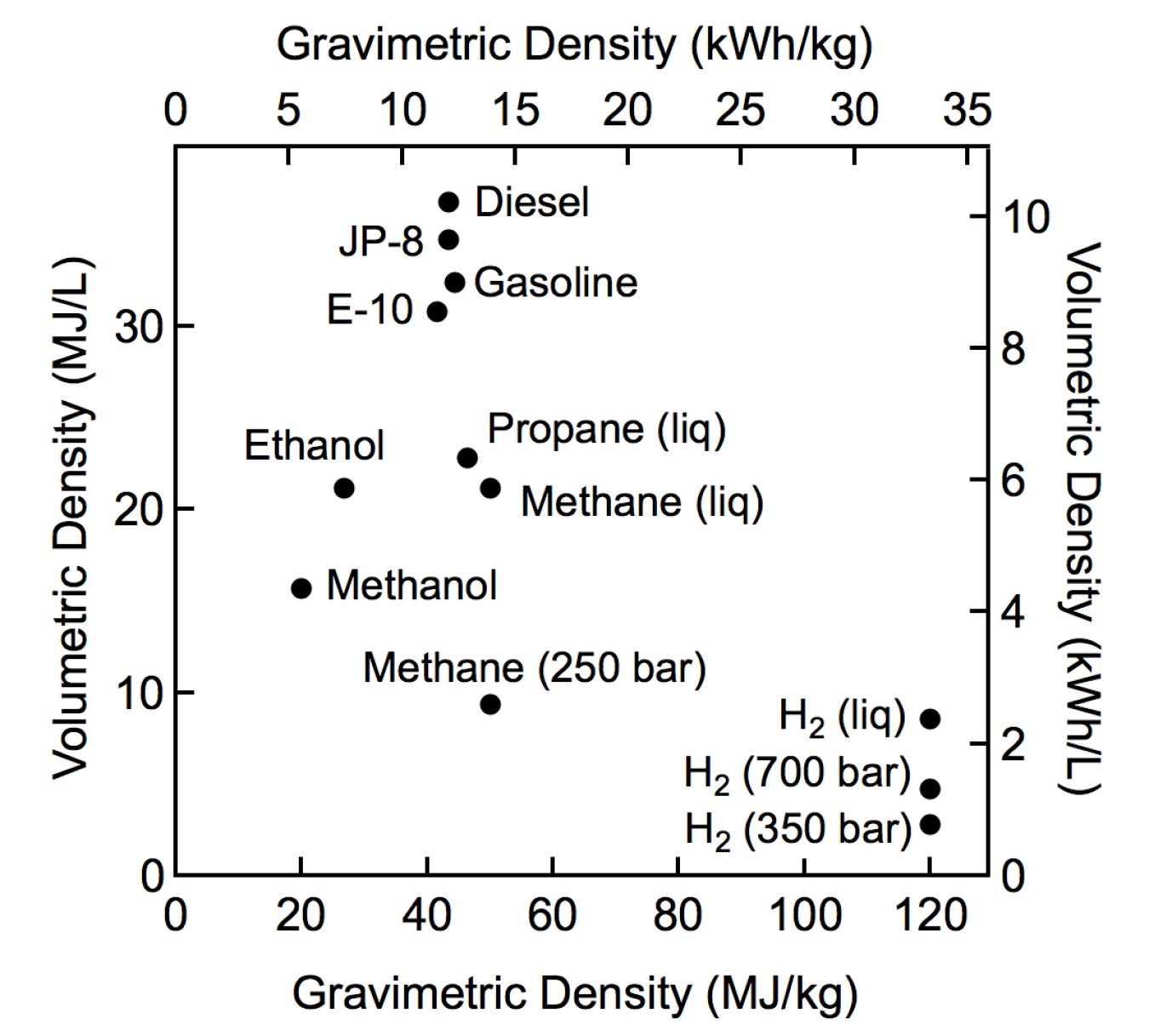
Fuel consumption and density (Autoweek, 2019) (Waterstof-info, 2019)... | Download Scientific Diagram

Chemical and Physical Solutions for Hydrogen Storage - Eberle - 2009 - Angewandte Chemie International Edition - Wiley Online Library
How many liters of hydrogen gas will be produced by the electrolysis of 5 liters of water? How do you write it down and explain it? - Quora

Processes | Free Full-Text | Recent Progress Using Solid-State Materials for Hydrogen Storage: A Short Review | HTML

Specific heat of one mole of hydrogen at constant pressure and at constant volume are 450 JK^-1 and 300 JK^-1 respectively. Then what is the density of the gas at S.T.P ? ( Patm = 1.013 × 10^5Nm^-2 )


![Steam methane reforming: inputs and outputs to produce 1 kg hydrogen [41]. | Download Table Steam methane reforming: inputs and outputs to produce 1 kg hydrogen [41]. | Download Table](https://www.researchgate.net/profile/Renata-Bura/publication/282393135/figure/tbl1/AS:667926071894032@1536257393402/Steam-methane-reforming-inputs-and-outputs-to-produce-1-kg-hydrogen-41.png)